From left: Medical oncologist Neil Iyengar, radiation oncologist Amy Xu, and medical oncologist Joshua Drago were among many MSK physician-scientists who discussed the latest research at the San Antonio Breast Cancer Symposium this week.
This week at the annual San Antonio Breast Cancer Symposium (SABCS), MSK researchers discussed some of the latest advances in breast cancer treatment and led educational sessions about improving ways to treat and manage the disease.
Here are some of the noteworthy presentations involving MSK investigators:
Tailoring Diet and Exercise
Medical oncologist Neil Iyengar is a leading expert on the relationship between cancer, obesity, nutrition, and exercise. As part of an educational panel, he discussed a new trend toward personalizing diet and exercise recommendations, both to reduce risk for the disease and to improve response to treatment.
“A lot of recent research suggests we can be more specific about how to change our diets and the exercise that people should practice based on features of their cancer and other biological factors,” he explains.
A study led by Byuri Angela Cho of Weill Cornell Medical College and coauthored by Dr. Iyengar and other MSK colleagues reported that obesity can completely transform the gene expression of the breast cancer microenvironment — the tissues, blood vessels, immune cells, and other noncancerous components surrounding a tumor. The findings, reported in Carcinogenesis in October 2021, showed that when a patient is obese, genes in the microenvironment are turned on that promote tumor growth. Other genetic changes can trigger inflammation and prevent immune cells in the area from attacking the tumor.
“Based on emerging data, we are testing whether a whole-food, plant-based diet would reduce inflammation and reduce risk of recurrence for estrogen receptor-positive breast cancer,” he says.
Dr. Iyengar also discussed an ongoing trial at MSK testing the use of a ketogenic diet (which is very low in carbohydrates) in people whose breast tumors have a PI3K mutation. Drugs called PI3K inhibitors are used to treat these cancers, but they often cause high blood sugar and trigger an insulin spike that seems to undermine the drug’s effect. A study in mice suggested that a ketogenic diet, which reduces insulin, could improve the effectiveness of PI3K inhibitors.
“It’s becoming pretty clear we can directly affect tumor biology, the microenvironment, and response to cancer therapy with lifestyle interventions,” Dr. Iyengar says. “I think the problem has been that we’ve had recommendations that are generic: ‘Eat healthy food. Exercise is good.’ Now the data suggest we can be more precise, possibly prescribing exercise and diet in very specific ways.”
Host Diane Reidy-Lagunes speaks with Neil Iyengar, a breast medical oncologist and MSK’s leading researcher on diet, metabolism, and cancer.
Pinpoint Radiation for Metastatic Breast Cancer
Radiation oncologist Amy Xu was part of an educational panel discussing treatment of advanced breast cancer with local therapies (treatments directed to a limited area). Effective systemic therapies such as chemotherapy and targeted drugs are allowing people with breast cancer to live much longer, so there is now an increased focus on treating oligometastatic cancer — disease that has just begun to spread (metastasize) and is detected in a limited number of sites in the body.
MSK has a dedicated program to treat metastatic disease in breast cancer patients, led by radiation oncologists Atif Jalees Khan and Jonathan Yang. A key component of this program is targeted, high-dose radiation called stereotactic body radiotherapy (SBRT). The total radiation dose can be split into fewer fractions of larger doses given over a shorter period — an approach known as MSK PreciseTM. Dr. Xu says that improvements in image guidance, combined with treatment-planning software developed at MSK called ECHO, has made it possible to deliver SBRT with a high level of precision that reduces side effects and produces better outcomes for patients.
MSK radiation oncologists have demonstrated that SBRT can alleviate symptoms and change the course of the disease. In August 2021, Drs. Xu and Yang published results in Cancer Medicine showing that SBRT could provide long-term disease control and survival for people with oligometastatic breast cancer. Another study, reported in October 2021 by radiation oncologist C. Jillian Tsai, showed that SBRT significantly increased progression-free survival in oligometastatic lung cancer patients.
At the meeting, Dr. Xu discussed these findings and provided insight into where the research is headed.
“In the past, radiation for stage 4 breast cancer was limited to relieving symptoms, such as pain control, and even then, using low doses to minimize toxicity,” Dr. Xu says. “We’re now using radiation to control disease before it causes symptoms and prevent the cancer from spreading, potentially leading to cure. A lot more work is needed to identify which metastatic patients benefit from aggressive local therapies. It’s really exciting to be working on this frontier.”
Clarifying Resistance to HER2 Drugs

Emanuela Ferraro
A major advance in breast cancer treatment has been the emergence of drugs that treat HER2-positive tumors, which account for about 20% of breast cancers. These drugs, starting with trastuzumab (Herceptin®) in the 1990s, block the activity of HER2, a receptor on the surface of cells. This prevents the cells from receiving growth signals to fuel a tumor. However, most HER2-positive cancers eventually stop responding to these drugs and begin growing again.
Emanuela Ferraro, a postdoctoral fellow in MSK’s Breast Medicine Service and Human Oncology and Pathogenesis Program, presented new findings suggesting how some breast cancers develop this resistance to anti-HER2 therapies. A study led by physician-scientist Sarat Chandarlapaty and coauthored by Dr. Ferraro, medical oncologist Pedram Razavi, graduate student Alison Smith, and others, has identified genetic mutations that arise after the cancer cells are treated with these drugs.
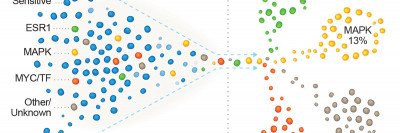
“We knew that there is a subset of patients less sensitive to anti-HER2 drugs, such as patients with tumors that have mutations in a pathway called PI3K,” Dr. Ferraro says. “But these mutations seem to be not enough to explain all cases of resistance to the drugs. We wanted to explore the effect of mutations in genes belonging to another pathway associated with HER2 signaling called MAPK.”
The team looked at the genetic makeup of 733 HER2-positive breast cancer samples. About half were from primary tumors and half were from metastases. They found that the cells that came from metastases had more frequent alterations in genes that activate the MAPK pathway, compared with the primary breast tumors — about 16% versus 10%. In addition, they looked at patients on first-line treatment (chemotherapy combined with anti-HER2 drugs) and showed that those with MAPK mutant tumors had a worse prognosis. Based on that, they hypothesized that alterations in the MAPK pathway could be a therapeutic target.
To test this theory, the research team used drugs called MEK/ERK inhibitors (which are already approved for treating people with melanoma) to treat the resistant cancer cells containing MAPK pathway mutations in a lab dish and in rats. They found that the cells were especially vulnerable to these drugs while still resistant to anti-HER2 agents. The results were published November 18 in Nature Communications.
“This is exciting because it’s the first time that this mechanism of HER2 signaling resistance has been described,” Dr. Ferraro explains. “The finding opens the possibility of a new therapeutic approach to test in clinical trials for this subset of patients with poor prognoses.”
A study led by MSK surgeon Andrea Barrio examined if race and ethnicity play a role in who is most likely to develop lymphedema, a side effect from breast cancer surgery.
Boosting HER2 Drugs with Statins
Medical oncologist Joshua Drago reported intriguing findings suggesting that statins, which are widely used to lower cholesterol, might make HER2-targeted cancer-fighting drugs work better.
“HER2 is expressed in the cell membranes, which are made of lipids,” Dr. Drago says. “A statin, which targets lipid metabolism, can potentially have a big effect on HER2 expression. Higher levels of HER2 could help a drug like trastuzumab bind better and be more effective.”
Earlier research by the laboratory of MSK radiochemist Jason Lewis studying HER2-positive stomach cancer cells found that a protein called CAV1 affects how HER2 is distributed through a cell, including how much is expressed on the membrane. When CAV1 amounts are lowered using statins, the HER2 protein stays longer on the cell membranes — presenting a better target for drugs such as trastuzumab.
Dr. Drago’s team investigated whether this held true in breast cancer cells for a HER2-targeted drug called T-DM1 (Kadcyla®). This is a drug consisting of trastuzumab and chemotherapy. “T-DM1 still binds to the HER2 on cancer cells, but it carries chemotherapy molecules with it like a Trojan horse. The drug is taken in by the cancer cells and destroys them,” he says.
The researchers studied the question in mouse models of breast cancer and in breast cancer patients. In the mice, they found that giving statins depleted CAV1, increased HER2 levels on the cell surface, and improved the effectiveness of T-DM1. In breast cancer patients who had taken statins and received T-DM1, medical records suggested that they might have longer progression-free survival (the time in which the disease didn’t worsen) compared with those who had not taken statins.
The group of patients studied was too small to draw any conclusions, Dr. Drago says, so he is currently studying the question in a much larger group of breast cancer patients.
Dr. Drago says that manipulating the presence of a drug target on cancer cells is a new approach to making drugs more effective. “Statins are protective against heart disease, and cardiac dysfunction is a known side effect of some HER2-targeted breast cancer therapies, so that could be another potential upside.”
- Diet and exercise plans may become more personalized for breast cancer.
- Powerful, precise radiation can control and possibly cure breast cancer that has spread.
- A new therapeutic target is identified in breast cancer that resists anti-HER2 drugs.
- Statins have the potential to make HER2-targeted therapies work better.